 The pain associated with the Aδ fibers can be associated to an initial extremely sharp pain. The first phase is mediated by the fast-conducting Aδ fibers and the second part due to (Polymodal) C fibers. This is due to the light or non-myelination of the axon. These only conduct at speeds of around 2 meters/second. The other type is the more slowly conducting C fiber axons. They are myelinated and can allow an action potential to travel at a rate of about 20 meters/second towards the CNS. Nociceptors have two different types of axons. Other nociceptors respond to none of these modalities (although they may respond to stimulation under conditions of inflammation) and are referred to as sleeping or silent. Some nociceptors respond to more than one of these modalities and are consequently designated polymodal. The majority of nociceptors are classified by which of the environmental modalities they respond to. Only when the high threshold has been reached by either chemical, thermal, or mechanical environments are the nociceptors triggered. The sensory specificity of nociceptors is established by the high threshold only to particular features of stimuli. This leads to the train of events that allows for the conscious awareness of pain. When the electrical energy reaches a threshold value, an action potential is induced and driven towards the central nervous system (CNS). The peripheral terminal of the mature nociceptor is where the noxious stimuli are detected and transduced into electrical energy. There currently is a lot of research about the differences between nociceptors. On the contrary, the peptidergic nociceptors continue to use TrkA, and they express a completely different type of growth factor. This transition is assisted by runt-related transcription factor 1 (RUNX1) which is vital in the development of nonpeptidergic nociceptors. The nonpeptidergic nociceptors switch off the TrkA and begin expressing RET proto-oncogene, which is a transmembrane signaling component that allows the expression of glial cell line-derived neurotrophic factor (GDNF). 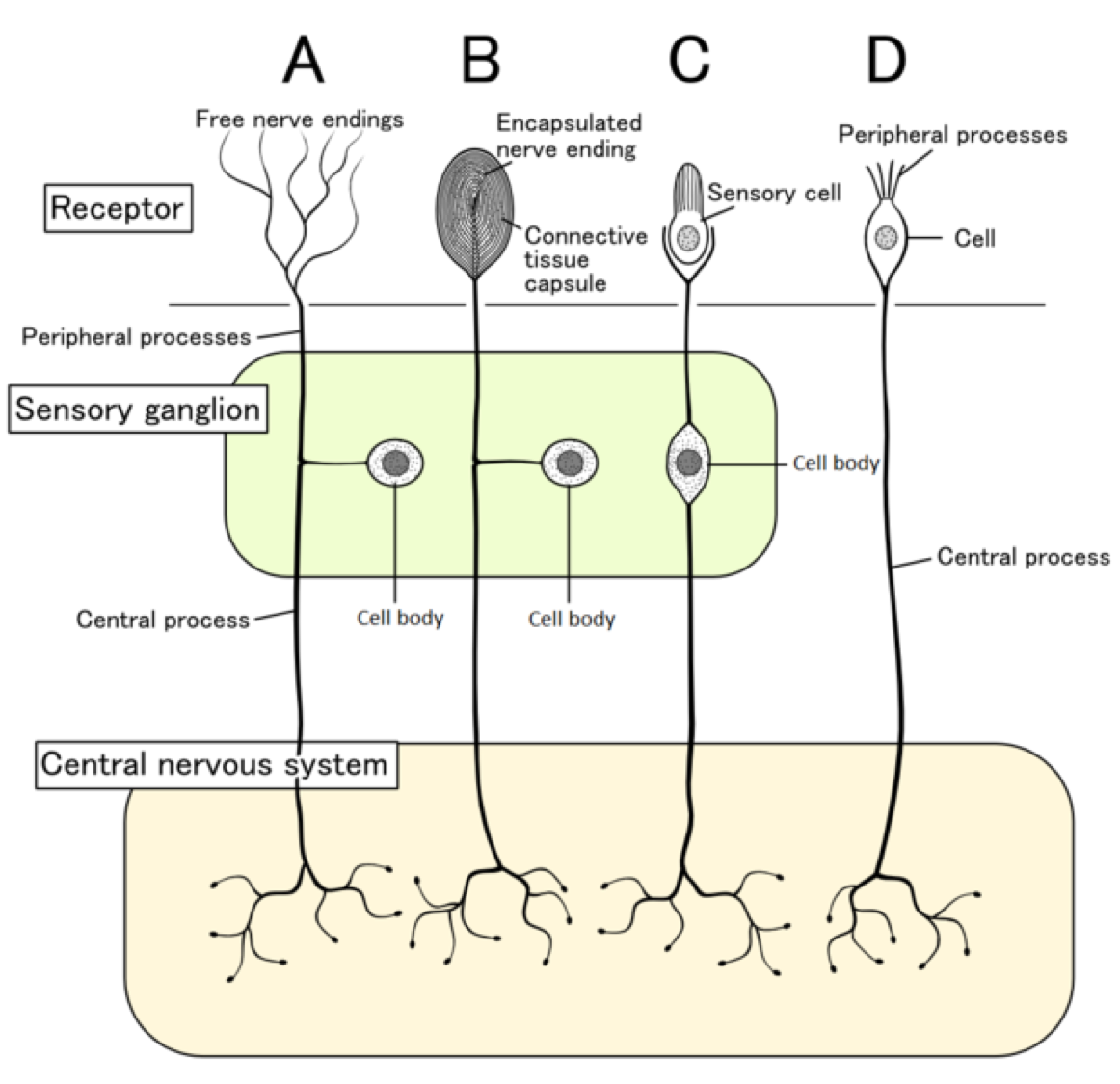
This differentiation occurs in both perinatal and postnatal periods. Their specializations allow the receptors to innervate different central and peripheral targets. They are classified as either peptidergic or nonpeptidergic nociceptors, each of which express a distinct repertoire of ion channels and receptors. įollowing sensory neurogenesis, differentiation occurs, and two types of nociceptors are formed. However, transcription factors that determine the type of nociceptor remain unclear. All neurons derived from the neural crest, including embryonic nociceptors, express the tropomyosin receptor kinase A (TrkA), which is a receptor to nerve growth factor (NGF). Earlier forming cells from this region can become non-pain sensing receptors, either proprioceptors or low-threshold mechanoreceptors. The neural-crest stem cells split from the neural tube as it closes, and nociceptors grow from the dorsal part of this neural-crest tissue. It is specifically responsible for development of the peripheral nervous system (PNS). The neural crest is responsible for a large part of early development in vertebrates. Nociceptors develop from neural-crest stem cells. The axons extend into the peripheral nervous system and terminate in branches to form receptive fields. The trigeminal ganglia are specialized nerves for the face, whereas the dorsal root ganglia are associated with the rest of the body. The cell bodies of these neurons are located in either the dorsal root ganglia or the trigeminal ganglia. Internal nociceptors are found in a variety of organs, such as the muscles, the joints, the bladder, the visceral organs, and the digestive tract. External nociceptors are found in tissue such as the skin ( cutaneous nociceptors), the corneas, and the mucosa. In mammals, nociceptors are found in any area of the body that can sense noxious stimuli. The specific receptors for these intense stimuli were called nociceptors. 
Some intense stimuli trigger reflex withdrawal, certain autonomic responses, and pain. Sherrington used many different experiments to demonstrate that different types of stimulation to an afferent nerve fiber's receptive field led to different responses. In earlier centuries, scientists believed that animals were like mechanical devices that transformed the energy of sensory stimuli into motor responses. Nociceptors were discovered by Charles Scott Sherrington in 1906. The brain creates the sensation of pain to direct attention to the body part, so the threat can be mitigated this process is called nociception. A nociceptor ("pain receptor" from Latin nocere 'to harm or hurt') is a sensory neuron that responds to damaging or potentially damaging stimuli by sending "possible threat" signals to the spinal cord and the brain. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
